S1.3.3, S1.3.4 & S1.3.5 – Electronic configuration
📌 Electronic configuration
- Electronic configuration shows how electrons are arranged in an atom
- Electrons occupy energy levels around the nucleus
- Energy levels are divided into sublevels and orbitals
📌 Energy levels and sublevels
- Energy levels are numbered 1, 2, 3, and 4
- Each energy level contains sublevels
- Sublevels are labelled s, p, d, and f
| Sublevel | Number of orbitals | Maximum electrons |
|---|---|---|
| s | 1 | 2 |
| p | 3 | 6 |
| d | 5 | 10 |
📌 Order of filling
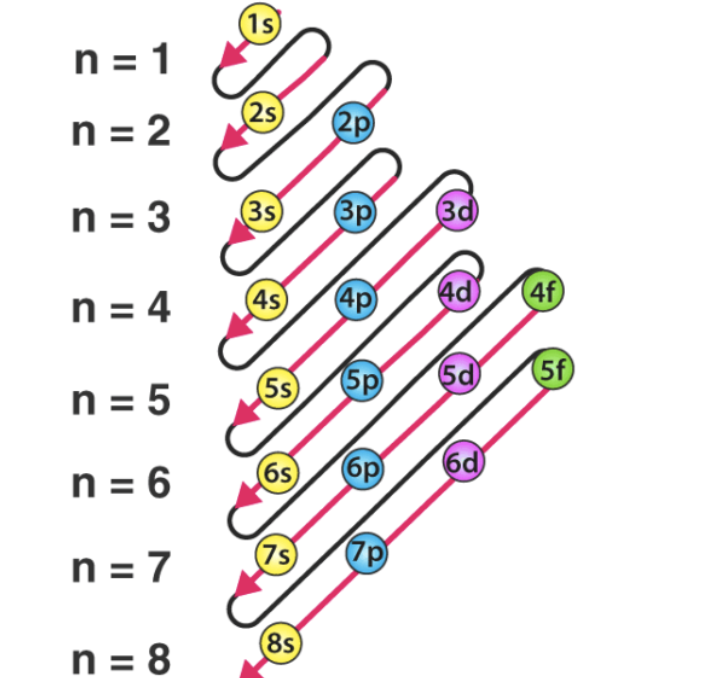
1s → 2s → 2p → 3s → 3p → 4s → 3d
📌 Rules for filling orbitals
- Each orbital holds a maximum of two electrons
- Orbitals of lower energy fill first – Aufbau principle
- Electrons in the same orbital have opposite spins – Pauli’s exclusion principle
- Orbitals of equal energy fill singly before pairing – Hund’s rule
EXAMPLE
Oxygen has 8 electrons, 2 in 1s 2 in 2s and 4 in 2p
1s orbital : ↑↓ (lowest orbital first fills with two oppositely spinning electrons)
2s orbital : ↑↓ (next orbtial fills with two more oppositely spinning electrons)
2p orbital: ↑↓ ↑ ↑ (p orbital singly fills with 3 same spinning electrons and one oppositely spinning one after being fully singly filled)
therefore the electronic configuration of oxygen is 1s² 2s² 2p⁴