R3.4.11 – Electrophilic addition reaction mechanisms (HL)
📌 Breaking of the pi bond
- Addition reactions involving alkenes require the breaking of the pi bond to create two single (sigma) bonds
- From information learnt in S2, we know that the pi bond is electron dense
- This electron dense area attracts electrophiles so that alkenes can undergo addition reactions
📌 Reaction mechanism
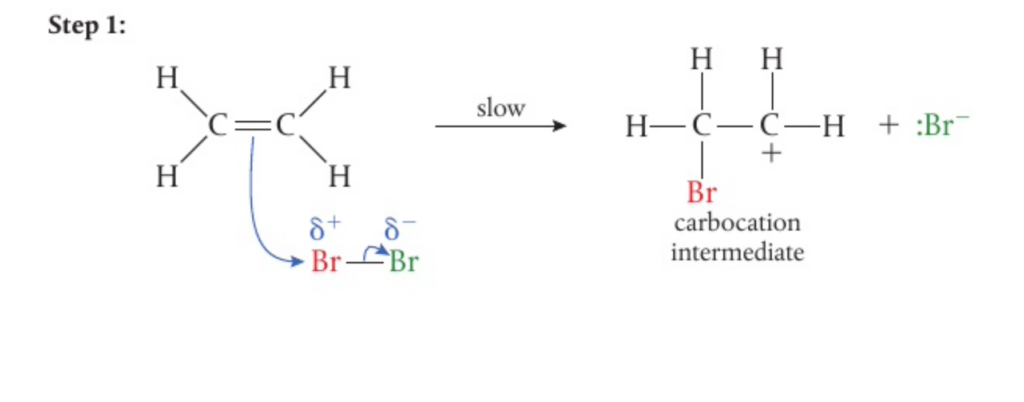
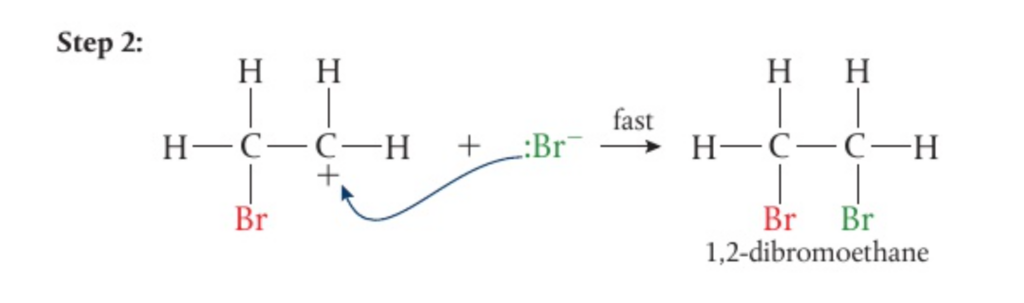
- Addition reactions involve a 2-step mechanism
- First, the electrophile undergoes heterolytic fission, following which the partially positive ion is attracted to the electron dense area of the pi bond
- This then creates an intermediate carbocation and a negative ion
- The negative ion is then attracted to the positive charge on the central carbon of the carbocation
- This reaction mechanism works for both halogens and hydrogen halides
- In the case of hydrogen halides, the hydrogen atom will always attach first, followed by the halogen
📌 Addition of water
- The addition of water can only occur in the presence of a strong catalyst as water is a weak electrophile
- First, the formation of a H3O+ ion which acts as a catalyst (any strong acid)
- The heterolytic fission of the H3O+results in the formation of a carbocation intermediate and a single water molecule
- Following this, the water molecule acts as a nucleophile and bonds to the intermediate carbocation