R3.3.2 – Homolytic fission
📌 Breaking covalent bonds
- Radicals can be created by the breaking of covalent bonds
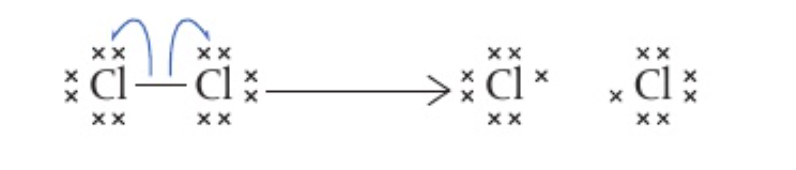
- Homolytic fission is the process by which a covalent bond breaks to form 2 identical radicals
- ‘Identical radicals’ suggests that both radicals have equal electrons assignment
📌 Types of fission
- Thermolytic fission : when sufficient energy for breaking bonds can be gained from heating
- Photolytic fission : when energy is gained from high-energy UV light for bond breaking
📌 Representing fission
- Single headed curly arrows (‘fish hook arrows’) are used to demonstrate the moving of electrons during homolytic fission
📌 Ozone depletion
- Chlorine radicals cause ozone depletion
- CFCs – chlorofluorocarbons – are compounds in aerosols, refrigerators etc and they release chlorine radicals when exposed to high-energy UV
- These radicals catalyse the decomposition of ozone (O3)