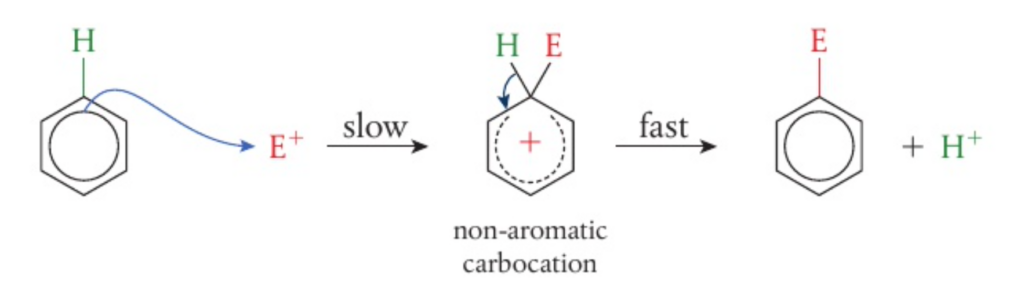
R3.4.13 – Electrophilic substitution of benzene
📌 Resonance
- Benzene is highly unsaturated but does not react like other alkenes (or unsaturated hydrocarbons)
- Due to it’s high stability and ring structure, benzene undergoes substitution rather than addition (as is the case with most double bond compounds)
📌 Electrophilic substitution
- The delocalised cloud of electrons attracts electrophilic species
- The hydrogen atom on one of the six carbon atoms can be substituted by the electrophile
- When an electron pair from the benzene is attracted to the electrophile, it causes a disruption in the delocalised pi bond system
- Following this, an intermediate compound is formed where both the leaving hydrogen and the electrophile are bonded to the benzene ring. This is represented by a dotted semicircle inside the ring compound
- After this, the hydrogen leaves the compound and the electrophile is substituted in its place
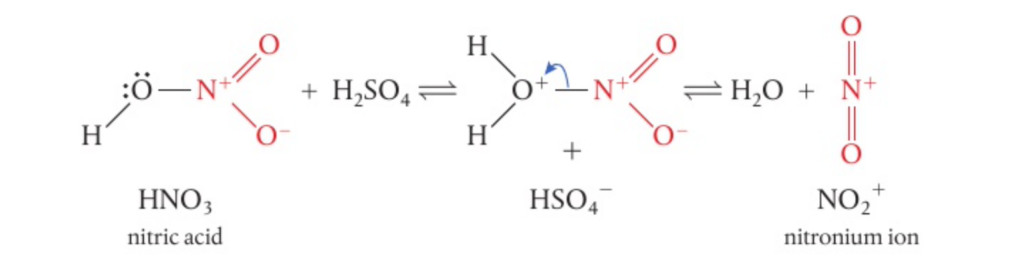
📌 Nitration of benzene
- 0One important reaction involving the electrophilic substitution of benzene is the nitration reaction
- This involves the substitution of NO2+ into the ring compound
- First, the nitronium ion is generated using the nitrating mixture of concentrated sulphuric acid and concentrated nitric acid at 50°C
- Once the nitronium ion is generated, it undergoes the same reaction mechanism as seen above to form nitrobenzene