R3.4.9 & R3.4.10 – SN1 and SN2 nucleophilic substitution mechanisms (HL)
📌 Types of halogenoalkanes
- Halogeonalkanes can be divided into 3 subgroups : primary, secondary and tertiary. The iB syllabus focuses on the reaction mechanisms of primary and tertiary halogeonalkanes
- Primary : Primary halogeonalkanes have at least 2 hydrogens attached to the carbon with the C-X bond (X is a halogen). These compounds undergo SN2 reactions which are substitution reactions involving nucleophiles (S is for substitution, N is for nucelophilic)
- Tertiary : Tertiary halogenoalkanes have three alkyl groups attached to the carbon with the C-X bond. These compounds undergo SN1 reactions
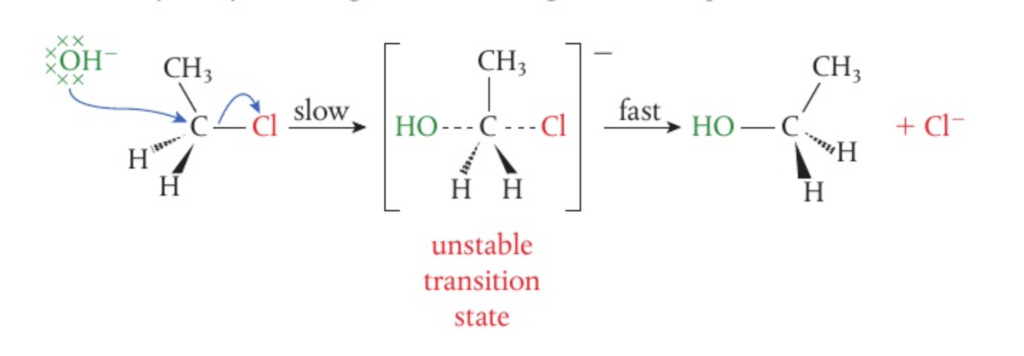
📌 Understadning SN2 reactions
- In such reactions, the C-X bonds breaks heterolytically, causing the halogen ion to be released. Additionally, the carbon takes on a slightly positive charge which causes the nucleophile to be attracted to it.
- This then forms an unstable intermediate state wherein both the nucleophile and the halogen are attached to the central carbon. The C-X bond then fully breaks and the halogen ion is released /
- Because the reactions depends on the concentrations of both the nucleophile and the halogenoalkane, SN2 reactions are bimolecular (hence the 2), meaning that they are second order overall
- Note that the nucleophile attaches itself on the opposite side from the halogen which causes an inversion of arrangement of atoms/groups around the central carbon. This is why SN2 reactions are considered to be stereospecific.
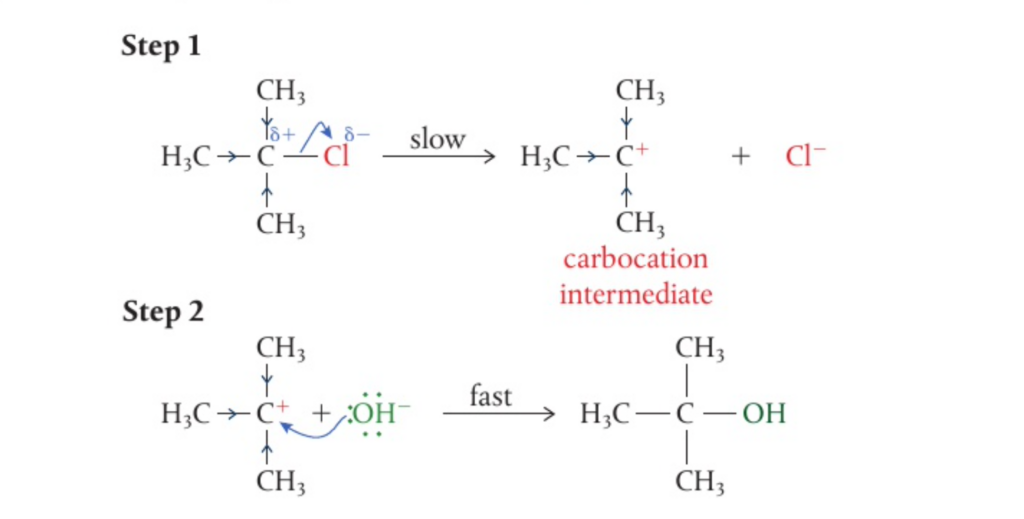
📌 Understadning SN1 reactions
- In such reactions, the alkyl groups around the central carbon cause steric hinderance
- Unlike in an SN1 reactions, when the C-X bond breaks heterolytically here, the halogen ion becomes the leaving group immediately and an intermediate cation is formed
- This intermediate is known as a carbocation and becomes easily attracted to the nucleophile
- Such reactions depend only on the concentration of the halogenoalkane, making it unimolecular (hence the 1)
- These reactions are no stereospecific and they demonstrate the idea of ‘positive inductive effect’ due to the 3 alkyl groups which stabilise the compound
📌 Halogens
- The quality of the leaving group depends on the strength of the C-X bond
- Greater bond strength means less energetically favourable for the bond to be broken
- The leaving groups can be ranked as the following : I– > Br– > Cl– > F– with iodoalkanes being the best and fluoroalkanes being the worst