S2.4 From Models to Materials
S2.4.1 The Bonding Triangle and S2.4.2 Applications of the Bonding Triangle :
- Bonding within a substance is best described as a continuum between ionic, covalent, and metallic bondinG
- This can be visualized through a bonding triangle
🔍TOK Connect : To what extent do the classification systems we use in the pursuit of knowledge affect the conclusions that we reach?
📌 Van Arkel-Ketelaar Bonding Triangle :
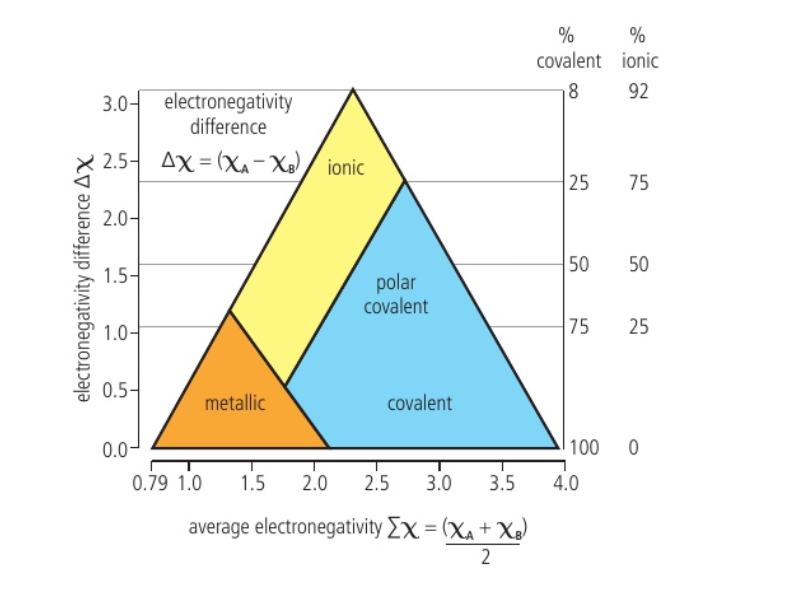
- The position of an element or compound in the bonding triangle is determined from its electronegativity values
- On the Y axis – calculate the difference in electronegativities
- On the X axis – calculate the average electronegativity
- Small difference in electronegativity, low average electronegativity – metals
- Small difference in electronegativity, high avg electronegativity – covalent
- Large electronegativity difference and mid-average electronegativity – ionic
- Model of a bonding continuum helps us make more accurate predictions of properties of compounds
- Limitations
- Bonding in Cl2 is not more metallic than in F2
- Does not predict the properties of transition metals and their compounds well
S2.4.3 Alloys :
- Mixture of two or more metals/metal and non metals with no particular composition
- Alloys have enhanced properties compared to common metals
- Eg. Brass is an alloy of Copper and Zinc
- Stronger and harder than pure copper or zinc
- Naval brass has tin which enhances its resistance to corrosion
- Alloys are produced by adding one metal element to another in its liquid state so the atoms can mix
- The production of alloys is possible due to the non directional nature of the delocalized electrons and the fact that the lattice can accomodate ions of different sizes
- Due to the presence of differently sized ions – sliding of layers is prevented, making alloys stronger and more chemically stable than the metals they’re made of
⭐️ Alloys have no fixed composition and hence cannot be represented by a chemical formula.
S2.4.4 Polymers :
⭐️ Polymers are long chain molecules formed when monomers join together through covalent bonding.
- Polymers are also known as macromolecules as they are composed of thousands of atoms
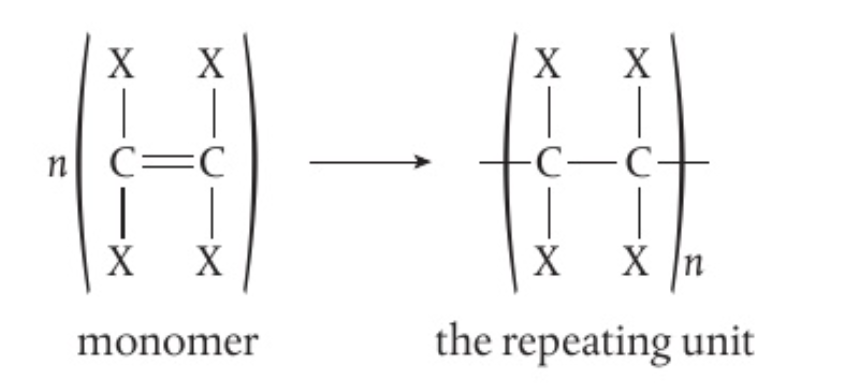
- Examples of natural polymers include proteins, DNA, starch
- Examples of synthetic polymers include polyethylene, polypropene
- Properties of plastics
- Strength and durability – PVC (polyvinyl chloride), nylon
- Flexibility – LDPE (low density polyethylene)
- Lack of reactivity – for storage containers
- Thermal insulation – low thermal conductivity, high specific heat capacity
- Electrical insulation
- Non biodegradable – stay in the environment for long periods of time
🌍 Real World Connection : Global production of plastics has risen exponentially since 1950. Much of the plastic produced ends up in the ocean where it destructively impacts marine ecosystems. It is estimated by 2030 that approximately 300 million tonnes of plastic could end up in the ocean.
- Biodegradable plastics may be plant-based eg made of corn
- Thermoplastics – have only IMF between two chains and hence can be repeatedly cooled and heated
- Strength of a polymer
- Length of chain (relative molecular mass)
- Degree of branching
- Arrangement of groups
- longer chains, less branching – stronger as increases strength of LDF
- Tensile strength – ability to resist a stretching force without breaking
- Hydrogen bonding increases strength – seen in Kevlar/Nylon
- Plasticisers increase flexibility
S4.4.5 Addition Polymers
- Addition polymers form by breaking a double bond in each monomer
- Alkenes join together to form polymers
- eg ethene as a monomer forms polyethene
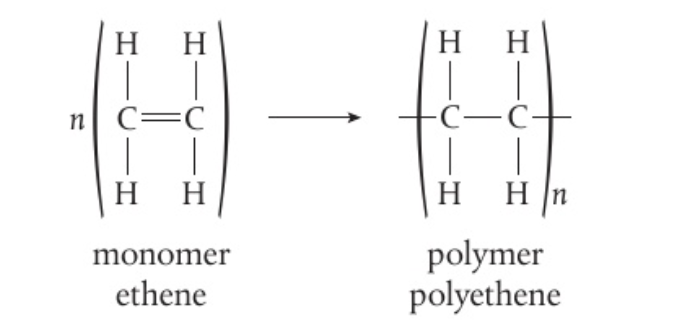
- Only C=C bond is affected
🔍 Exam Tip : Remember the polymer is named according to its monomer. Hence the polymer made from ethene monomers is called polyethene, even though the actual components are ethane (since the double bond is broken).
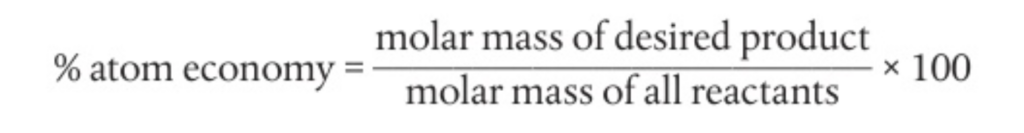
- Addition polymers do not generate a byproduct so convert 100% of reactants into product
S4.4.6 Condensation Polymers [HL]
- Condensation polymers form with a reaction between function groups of different monomers and the release of a small molecule like water
- In order to form condensation polymers, monomers must have two different functional groups (think of them as ends to react)
- Polyester
- Carboxylic acid and alcohol react to form polyester
- dicarboxylic acid + diol forms polyester (two functional groups)
- Number of water molecules in one less than the number of monomers produced
- Ethane-1,2-diol and Benzene-1,4-dicarboxylic acid = PET (Polyethylene terephtalate)
🔍 Paper 2 Tip : When you draw polymers, make sure to show open bonds on both sides of the bracket surrounding a monomer, so that you can show its ability to extend in both directions. See the image below :
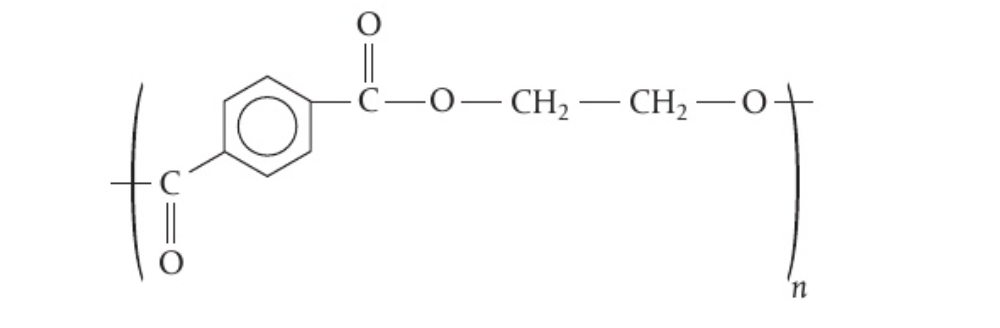
- Polyamide
- Carboxylic acid and amide react to form polyamide
- Polyamides also form when one monomer has an acid group and one has an amino group – amino acids
- Dicarboxylic acid + diamine = polyamide
- Used in textile industry – Kevlar, nylon
- Nylone 6,6 = 1,6-diaminohexane + hexandioic acid
- Hydrolysis breaks down polymers into monomers
- Uses water
- Occurs in presence of acids/alkalis/enzymes