S2.3 The Metallic Model :
S2.3.1 and S2.3.2 The Metallic Bond :
⭐️ Metallic bond is the electrostatic attraction between a lattice of cations and delocalised electrons
- Metals are found on the left side of the periodic table
- They have low ionization energies, enabling them to react with other atoms by donating their electrons and forming positive ions
- In elemental states, electrons are held loosely and are referred to as ‘delocalised’
- Metals contain a regular lattice arrangement of cations surrounded by a ‘sea’ of delocalised electrons
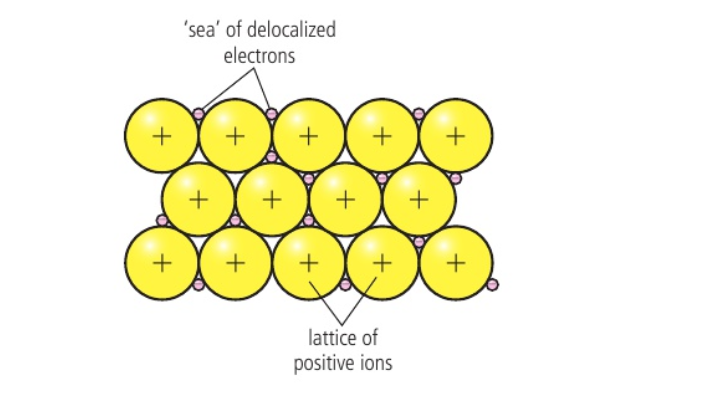
📌 Properties of Metals
- Physical properties
- Lustrous – delocalised electrons in crystal structure reflect light
- Good conductors of electricity (solid/liquid state) as electrons are free to move
- Good conductors of heat – delocalised electrons and packed structure of ions enable efficient thermal transfer
- Malleable and ductile – movement of delocalised electrons is non-directional so bond remains in place even under pressure
- High melting points
- Chemical properties
- Form cations
- Usually form ionic compounds
- Oxides are basic
- Non directional nature of metallic bonding allows for enhancement of properties through addition of different elements – results in the formation of alloys
- Steel is an example of an alloy of iron that contains small amounts of carbon
🔍 TOK Connect : The ability to explain natural phenomena such as metallic properties through the application of theory (eg. atomic theory) is an important feature of science.
📌 Strength of Metallic Bond
- Strength of a metallic bond depends on the charge and radius of metal ions
- Number of delocalised electrons
- Charge on the cation
- Radius of the cation
- Increased delocalised electron density and smaller cations result in strong bonds (Mg has stronger metallic bonding than Na)
- Strength of metallic bonding tends to decrease down a group as the size of cations increase
- Strength of metallic bonding tends to increase across a period due to increased charge density and smaller atomic radii
- Charge density = charge/volume
S2.3.3 Transition Metals [HL]
⭐️ Transition metals are elements that have incomplete d-subshells or can give rise to cations with incomplete d-subshells
- Transition metals have high melting points
- Close proximity in energy of 3d and 4s subshell – allows transition metals to delocalize a larger number of electrons
- All transition metals can delocalize electrons and have small ionic radii – increases the strength of metallic bonding
- Makes it harder to predict trends
- Sc to V – increase in mp due to increased number of delocalised electrons
- V – general decrease as the large amount of energy required to create +5 and +6 ions would not be paid back by the energy released by metallic bonding
- Transition metals have high electrical conductivity
- Large number of delocalised electrons allows for easy flow of current
- All metals can conduct electricity due to mobile electrons
- First row transition metals (except for Copper) seem to be poorer conductors of electricity compared to metals like Aluminium,etc
- s and p block electrons have a higher tendency to get delocalised than d block electrons – why most transition metals aren’t better electrical conductors than metals
- Copper has an extremely high electrical conductivity and is an exception – however, its conductivity is usually explained in terms of the delocalised s electron